|
Interestingly, one unique property of carbohydrates is that all monosaccharides with greater than five carbons can form either furanoses or pyranoses. Haworth projections of glucose (left) and fructose (right) For instance, biochemists classify five membered rings as furanoses and six-membered rings as pyranoses. Cyclization of glucoseīiochemists call the cyclic form of monosaccharides their Haworth projection, a carbohydrate diagram used to further classify sugars. This occurs in both aldoses and ketoses, resulting in a five or six-membered ring where one of the members is the oxygen from the attacking hydroxyl. With an acid catalyst, monosaccharides spontaneously undergo a cyclization reaction, where the carbonyl group is attacked by the hydroxyl of carbon 4 or 5. Glucose enantiomers: L-glucose (left) and D-glucose (right) Cyclizationĭespite the usefulness of the Fischer projection, most five or six-monosaccharides generally have cyclical molecular structures, due to unique carbohydrate structure and properties. Interestingly, biological systems only use one enantiomer of each monosaccharide, with D confirmation. Specifically, D sugars have the hydroxyl group pointing right, while L sugars have a left-pointing hydroxyl group. In the Fischer projection, the designation of D versus L depends on the direction of the hydroxyl group of the chiral carbon furthest from the carbonyl. Biochemists use the letters D and L to differentiate between monosaccharide enantiomers. This means that they have the same chemical formula, but different connectivity between their atoms.Īdditionally, many monosaccharides are enantiomers, or mirror images, of one another. Fructose structure Fischer Projection Glucose structure Fischer projectionīetween monosaccharides with the same number of carbons, aldoses and ketoses are what chemists call “ constitutional isomers” of one another. Contrastingly, monosaccharides with their carbonyl on an internal carbon are called ketoses, referencing the resultant ketone. Monosaccharides with their carbonyl at one end of the carbon chain are called aldoses, referencing the aldehyde group formed by the carbonyl. Isomersīiochemists classify monosaccharides differently based on the location of their carbonyl group. Fructose, a six-carbon monosaccharides Ribose, a five-carbon propertiesīiochemists number the carbons of monosaccharides based on the position of the carbonyl, with the end closest to the carbonyl labeled as carbon 1. When illustrated as a linear molecule (Fischer projection), monosaccharides have a carbonyl group attached to one carbon, while the other carbons have an alcohol group as a functional group. All monosaccharides have a chemical formula of C(n)H(2n)O(n), with a basic structure of an unbranching chain. In fact, monosaccharides are monomers of larger carbohydrates, meaning that they are the smallest unit of carbohydrate structure and properties, and form the building blocks of larger molecules. Monosaccharides are the simplest carbohydrate molecules. As we will see, carbohydrates have remarkable structural diversity. Their important functions in metabolism, structure, and identification are allowed by carbohydrate structure and properties. Carbohydrates also aid in cellular identification, as unique chains of sugars attached to a cell membrane provide a “fingerprint” that allows recognition of the type and species of the cell. Additionally, carbohydrates have structural functions as components of cell walls in plants and bacteria. Indeed, they play an important role in energy storage, in the form of polysaccharides, and energy expenditure, in the form of monosaccharides or “sugars”. 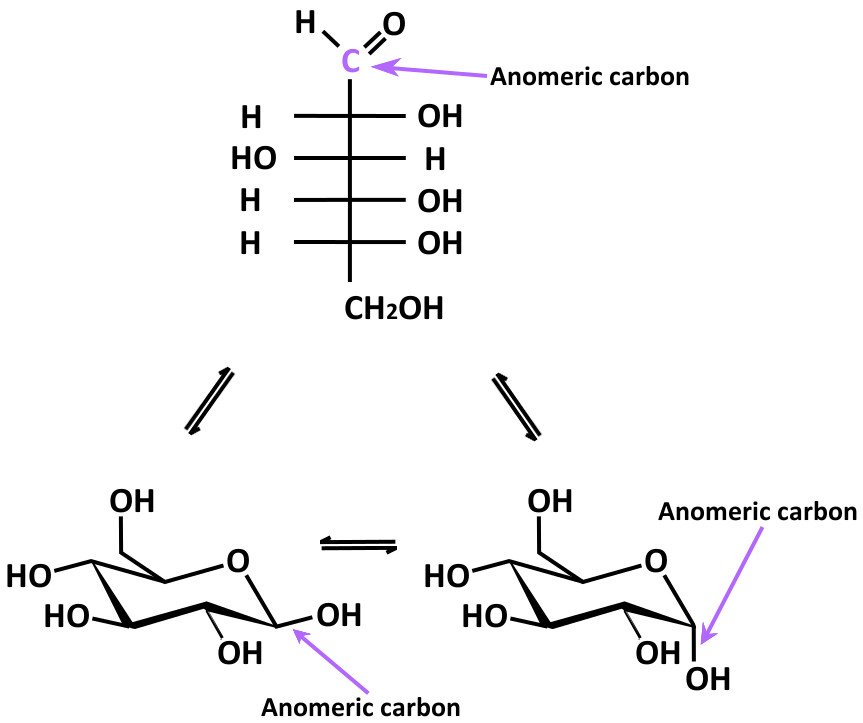
Specifically, carbohydrates are known for their functionality as units of energy, which is why “carbs” are an essential part of the human diet. Topics Covered in Other ArticlesĬarbohydrates are one of life’s most important classes of molecules. 
In this article, we will learn about carbohydrate structure and properties of the three main carbohydrate classes: monosaccharides, oligosaccharides, and polysaccharides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
